 Blogs
Blogs
In 2022 we had 42 FDA Drug Approvals and 3 were Gene Therapies using a viral vector. 2 of the approvals in 2022 were AAV based. The third is manufactured by adding functional copies of the transgene to the patient’s own Hematopoietic Stem Cells (HSCs)…
 Blogs
Blogs
Cell and Gene Therapies (CGTs) has an estimated market size value in 2022 of USD 8.22 billion and a revenue forecast in 2030 of USD 24.5 billion. This is a CAGR (compound annual growth rate) of 14.6% from 2022 to 2030. Needless to say, the…
 Webinars
Webinars
Being produced by Xtalks on Friday, June 24, 2022 | 12pm EDT (NA) / 5pm BST (UK) / 6pm CEST (EU-Central) 60 min Webinar Description: Cell and gene therapies (CGTs) are types of treatment that use cellular or genetic material with the goal of treating a disease or a…
 Blogs
Blogs
Dr. Sohrab Habibi Goudarzi – Senior Director of Pharma Discovery and R&D Services, Bryan Parmentier – Senior Principal Scientist of Pharma Method Development, and Yu-Hui (Ann) Fu -Principal Scientist of Pharma Regulatory, along with other scientists at KCAS Bioanalytical & Biomarker Services, coauthored with scientists at Gilead Sciences, Inc. in…
 Blogs
Blogs
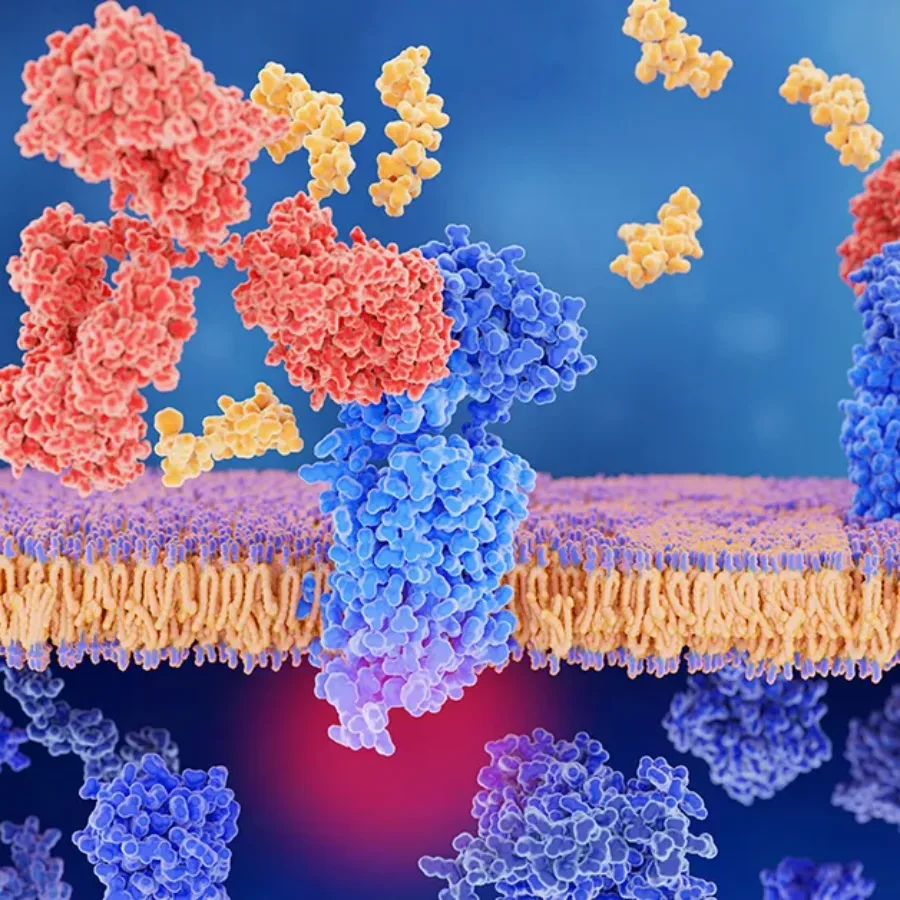
What are receptor occupancy assays?
 Blogs
Blogs
Tissue and Non-Liquid Matrices (such as cell lines, red blood cells, PBMCs, bone marrow aspirates, and suspension cells) are valuable matrices[] to test for biomarkers (PD) and drug levels (PK). Testing of tissues and…
 Blogs
Blogs
Pharmacodynamics (PD) is defined as the study of the biochemical and physiological effects of drugs and the mechanisms of their actions. Where pharmacokinetics looks at how the organism processes the drug, pharmacodynamics studies how…
 Blogs
Blogs
Drug discovery is an essential part of the drug development process. Testing in drug discovery is the mechanism to provide drug candidates that are selected for regulated animal studies and hopefully moving into clinical trials.
 Blogs
Blogs
At KCAS, our Discovery group performs bioanalytical / biomarker method development that can include any of the various components required for a validated method under the GLP guidelines. So why is Discovery group needed? The beginning stage of drug development is a stage of trial and error. It is generally…
 Blogs
Blogs
Finding a laboratory to successfully transfer and GLP validate your PK method can be a challenge. Yes sure the big labs can do it, but if you are not a preferred provider you may find yourself working with the B team that doesn’t move as fast as you need, and…