At KCAS Bio, we supported a therapeutic monoclonal antibody for advanced solid tumors from non-human primate (NHP) toxicology through first-in-human (FIH) dose escalation into phase 2 expansion. This project combined receptor occupancy assays (ROA) by flow cytometry for cell-surface binding on monocytes, immunophenotyping (IPT) for activation markers, and target engagement (TE) ligand binding assays (LBA) for soluble target in serum.
ROA and TE Fundamentals
Receptor Occupancy Assay (ROA): Flow cytometry method that quantifies free receptors (via labeled competing antibody or drug) and total receptors (via non-competing antibody) on cell surfaces like monocytes to calculate % occupancy by therapeutic antibody, supporting Pharmacokinetics/Pharmacodynamics (PK/PD) relationships (Liang et al., 2016).
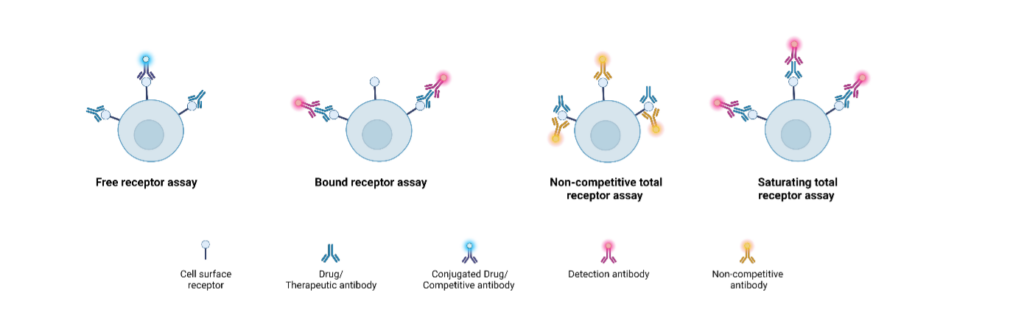
Target Engagement (TE) by LBA: ECLIA (ElectroChemiLuminescence ImmunoAssay) assays measuring soluble target in serum as free, bound (drug-target complex via anti-idiotype), or total (with excess drug), providing a proximal PD biomarker of drug binding for safety/PK (Ni et al., 2022).
ROA Evolution: Challenges and Solutions
A/ Nonclinical NHP Phase
Challenge: Remote in-life CRO required blood stabilization for shipping to central flow analysis; options needed to preserve RO signal over days.
Solution: The client requested ROA development for an NHP toxicology study. The in-life CRO was remote from our facilities, so we required blood preservation solutions. We conducted a pilot study on 3 NHPs with dose-response testing. This pilot tested three conditions:
- Lyse & fix then freeze and ship on dry ice; stain & acquire at KCAS.
- Stain then lyse & fix, freeze, and ship on dry ice; acquire at KCAS.
- Stain, lyse & fix, then ship at +5°C; acquire at KCAS.
We evaluated all conditions at 24 hours, 5 days, and 9 days post-processing. The preferred method was stain, lyse & fix, freeze, and ship on dry ice, with acquisition within 9 days to allow batching. The RO assay used free receptors detected with a labeled competing antibody. We processed over 500 samples that were stained in-life, frozen, shipped in 24 batches, and acquired on a flow cytometer within 9 days of staining/freezing.
B/ FIH Clinical
Challenge: The first-in-human (FIH) multi-arm dose-escalation study spanned multiple EU/US sites over more than 2 years with 24-48 hour transit times. Stabilizers worked for competing antibody (free receptors) but not non-competing antibody (total receptors), requiring fresh blood with room temperature stability up to 96 hours. Dual methods, RO plus immunophenotyping (IPT) measuring CD80, CD86, and HLA-DR on myeloid cells, supported phase 2 dose recommendation across 33 patients, 6 countries, and ~300 samples (2023-2026), leading to expansion phase recruiting additional subjects both in US and EU.
Solution: We pivoted to fresh blood, validated intra-/inter-run, day, and analyst precision, and confirmed 120-hour room temperature stability on in-study samples. We ran two methods on the same samples:
- ROA: Two tubes with non-competitive antibody for total receptors and fluorochrome-labeled drug for free receptors.
- IPT: CD80, CD86, HLA-DR on myeloid cells to assess activation status.
Close sponsor coordination ensured timely fresh blood logistics for the 33-patient, 6-country dose-escalation (~300 samples, 2023-2026).
C/ Phase 2
Challenge: Patient addition in multiple sites; Lyon-only instrument inadequate for multi-site operations.
Solution: Positive dose-escalation results led to expansion with additional patient at the US and EU. The initial methods had been developed on a Lyon-only instrument, so we transferred them to harmonized spectral cytometers (Cytek Aurora) across Lyon and Philadelphia. RO data from the escalation phase were sufficient. We redeveloped the IPT for spectral cytometry with panel optimization and an additional monocyte activation marker. We qualified the method in Lyon, cross-validated instruments and sites, and transferred it to Philadelphia, which has received the first expansion phase samples.
ROA Conclusion
From NHP tox to FIH escalation/expansion, ROA/IPT overcame logistics via stabilization pilots, fresh protocols, and spectral transfer, enabling global cell-specific PD tracking.
TE LBA Journey: Challenges and Solutions
Method Development
Challenge: We needed to quantify free and total soluble target in serum despite interference induced by the therapeutic drug. Initial sandwich formats failed despite multiple optimizations. This supported a parallel phase 1 target engagement (TE) trial with approximately 170 patients across 15-20 sites.
Step-by-step solutions:
1. Free Target Assay Challenge and Rationale
After selecting an appropriate recombinant protein for standards, we conducted several ECLIA sandwich tests for free target on the MESO QuickPlex SQ 120 from Meso Scale Discovery (MSD). These used a non-competitive capture antibody and a biotinylated detection antibody. The format failed because the detection antibody strongly competed with the therapeutic antibody for binding to free target, making accurate quantification impossible. We therefore shifted focus to total target measurement.
2. Initial Total Target Assay Challenge and Rationale
We tested six classical sandwich variations for total target, including sequential and non-sequential protocols, excess detection antibody, and non-competitive detection antibodies. None provided reliable performance across the expected ranges of therapeutic antibody and target concentrations in future clinical samples. This led us to abandon classical sandwich approaches for total target.
3. Bound Target Assay Challenge and Rationale
To overcome persistent issues, we changed strategy to directly measure the bound form, the target-therapeutic antibody complex, using antibodies targeting the therapeutic antibody rather than the target itself. We evaluated two detection options for the complex: anti-human IgG or anti-idiotype antibody. The anti-idiotype format delivered highly satisfactory results with minimal interference and good sensitivity.
4. Total Target Assay Adaptation Rationale
Building on the successful bound target anti-idiotype format, we adapted it for total target (free + bound) by adding excess therapeutic antibody during the assay. This ensured all free target formed detectable complexes, allowing consistent quantification of total target using the same core reagents and protocol as bound target.
5. Final Optimization and Implementation
We fine-tuned parameters, including antibody concentrations, incubation temperatures, diluents, saturation buffers, sample dilutions, and quality control preparation, to develop two robust ECLIA methods with a single calibration range but two different series of QCs (one for each assay). These reliably quantified total target and bound target (therapeutic antibody-complexed) in human serum across clinically relevant therapeutic antibody and target concentration ranges.
Evaluation of method performance
Challenge: We needed to ensure precision in frozen human serum despite matrix and drug interference effects while defining storage and transport stability.
Solution: The methods proved accurate and precise for both bound and total target with no matrix interference. Stability testing confirmed suitability for 3 freeze-thaw cycles, 72-168 hours at room temperature, and 6-12 months at -80°C.
Clinical Implementation: These dual ECLIA methods now support ongoing multicenter phase 1 safety and pharmacokinetic analysis.
TE Conclusion: The bound and total target ECLIA methods succeeded after abandoning free-target approaches, providing robust systemic target engagement data alongside receptor occupancy for comprehensive pharmacokinetics/pharmacodynamics understanding.
Complementary Insights Across Phases
| Aspect | ROA (Flow Cytometry) | TE LBA (ECLIA-MSD) |
| Matrix | Whole blood (fresh/stabilized) | Serum (frozen) |
| Readouts | Free/total cell-surface receptors | Bound/total soluble target |
| Specificity | Monocyte RO + activation (IPT) | Circulating target binding |
| Key Hurdles | Stabilization/shipping; instrument transfer | Assay interference; complex formats |
| Scale | >500 NHP; 300 FIH samples | ≥170 multicenter patients |
This project, from preclinical RO to clinical TE, Lyon to Philadelphia, demonstrated adaptive bioanalysis bridging cell-level modulation and systemic engagement to guide dosing in oncology development