Developing a robust Pharmacokinetic (PK) assay for biotherapeutics often presents unforeseen biological hurdles. Occasionally, a traditional Ligand Binding Assay (LBA) may yield inconsistent data. In this particular case, the LBA assay was not reliably detecting drug in the matrix, potentially indicating some matrix stability issue during method development. In addition, when mouse PK samples were analyzed, there appeared to be a “PK/PD disconnect”, where the drug appeared to be undetectable in the plasma despite the subject showing clear therapeutic efficacy. In this type of situation additional investigations are typically required, or alternative approaches are investigated.
In this blog, we’ll explore how pivoting from LBA to Hybrid LC-MS/MS allowed us to identify molecular instability, develop a successful PK assay, explain the “PK/PD disconnect”, and ultimately correlate drug concentration with efficacy.
The Challenge: Plasma Instability and A Disappearing Signal
The sponsor requested we develop a PK assay for therapeutic protein in mouse plasma. Initially, this assay was unsuccessfully attempted by LBA at a different contract research organization (CRO). The sponsor sought out KCAS Bio since we have expertise in several technologies that would allow us to further investigate this potential instability.
During the initial method development of the PK assay by LBA, the assay seemed to work sufficiently in buffer indicating that the reagents were able to bind the drug in a sandwich format, however when the drug was put into matrix, there was a loss of signal and inconsistent detectability.
- The Symptom: Drug in matrix was not detectable over time vs drug in buffer. Additionally, samples taken during the PK study showed no detectable drug.
- The Paradox: Despite the “lack” of drug detected, the animal subjects still exhibited efficacy (PD).
- The Hypothesis: The molecule was experiencing site-specific instability that prevented the ability of the LBA to detect it successfully in the typical “sandwich” format.
The Solution: Hybrid LC-MS/MS Multiplexing
To investigate this instability, the team pivoted to a Hybrid LC-MS/MS approach. Unlike LBA, which requires two binding events to generate a signal, LC-MS/MS can function with a single Immuno-Capture (IP) step and then monitor multiple specific peptides across the entire protein.
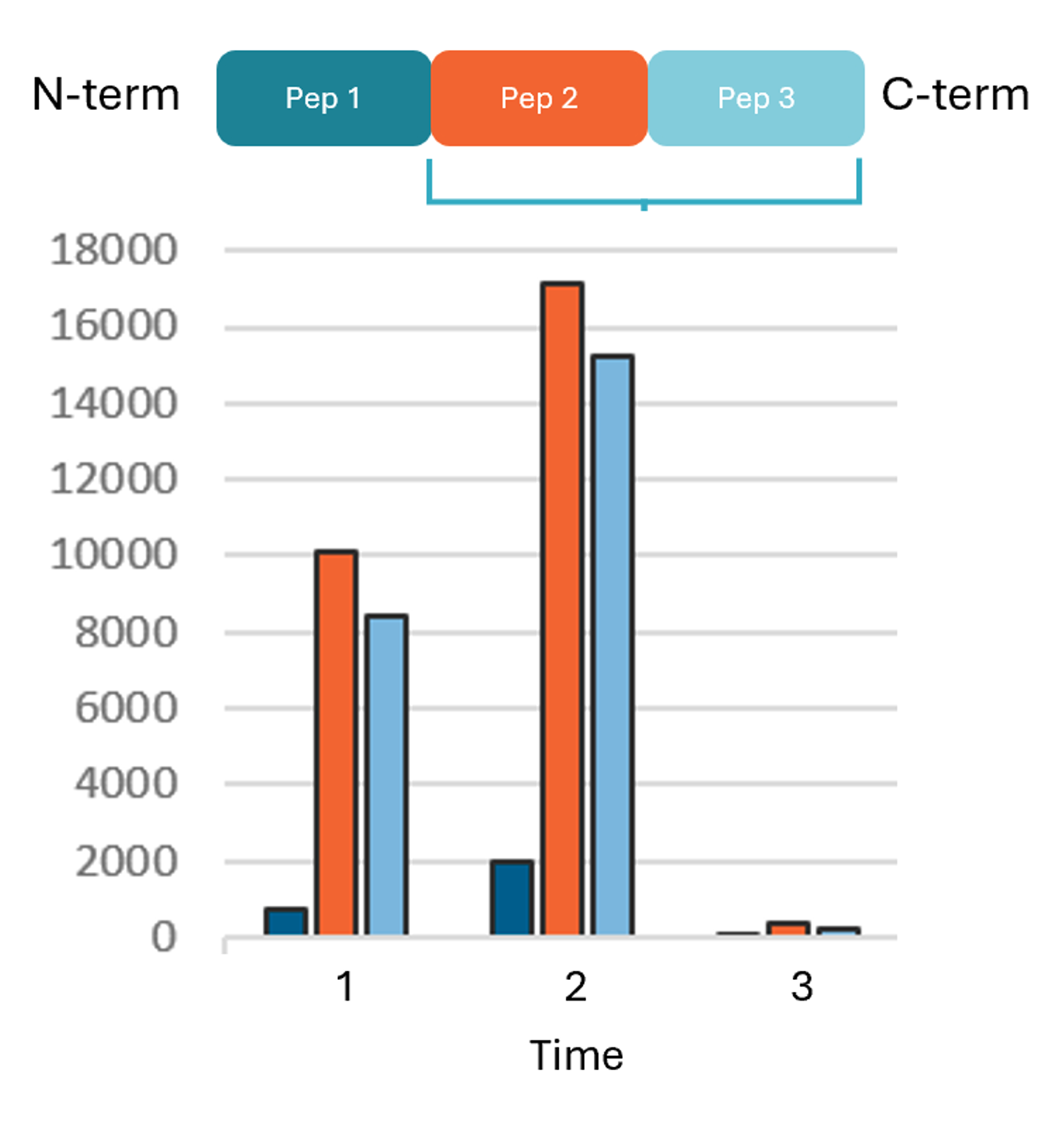
The team developed a single multiplexed assay to monitor three distinct regions of the molecule: which allowed us to ultimately monitor several parts of the protein simultaneously to see if this would shed any light into the instability observed.
- Peptide 1: Close to the N-terminus
- Peptide 2: Somewhere in the Middle region
- Peptide 3: Close to the C-terminus
Why Hybrid LC-MS/MS?
- The assay still needed good sensitivity but we wanted to use an orthogonal technology that might give us additional information into the problem.
- Reduced Reagent Dependency: It only requires one reagent (antibody) for capture, which is less likely to be affected by specific cleavage points. (i.e. if part of the molecule was being cleaved, we might still be able to capture, provided we capture the part that is still intact)
- Multiplexing Capabilities: It can track different parts of the molecule simultaneously in a single assay.
- Flexibility: We can “flip” the capture and detector antibody to different ends of the molecule to hopefully allow the capture of the drug. In contrast, the LBA assay would need both epitopes intact for a successful assay.
The Outcome: Identifying the Cleavage Point
By analyzing the time course of the PK study, the LC-MS/MS data provided the definitive evidence needed to understand the assay failure:
| Time Point | N-Terminus (Pep 1) | Middle (Pep 2) | C-Terminus (Pep 3) |
| Early | Reduced detection | Present | Present |
| Mid | Absent | Present | Present |
| Late | Absent | Cleared | Cleared |
The data proved that the N-terminus was being cleaved almost immediately in the PK study. Because the LBA likely relied on an N-terminal antibody for detection or capture, the assay failed as soon as that piece fell off.
However, because the middle and back of the molecule remained intact and present, the drug remained active. This successfully explained the PK/PD disconnect: the N-terminus was not required for therapeutic efficacy.
Lessons Learned for Biotherapeutic Development
This highlights Hybrid LC-MS/MS as a powerful alternative when LBA methodology hits a wall.
- Stability Insight: LC-MS/MS provides additional structural stability information that a standard LBA typically cannot. In addition, the LC-MS/MS assay is less reliant on the critical reagent and can “dial” in the needed selectivity to give additional information across the entire molecule.
- Validated Success: The resulting LC-MS/MS method was successfully validated across several species.
- Strategic Pivoting: When an assay fails, switching methodologies can provide the “additional information” needed to understand the biology of your therapeutic.
When your biotherapeutic project encounters unexpected results like a PK/PD disconnect, KCAS Bio partners with you to pivot strategically, moving beyond standard methodologies to find the right solution. Reach out to the KCAS Bio team to learn how our adaptive expertise can ensure your project’s success, no matter the analytical challenges.
Frequently Asked Questions
What is hybrid LC-MS/MS in bioanalysis?
Hybrid LC-MS/MS combines immunocapture with mass spectrometry to quantify multiple peptides from different regions of a protein therapeutic simultaneously.
How does multiplex peptide monitoring improve PK analysis?
Tracking multiple peptides provides region-specific information that helps identify structural degradation while maintaining accurate drug quantitation.
Can hybrid LC-MS/MS replace ligand binding assays?
Hybrid LC-MS/MS is often complementary rather than a replacement. Many development programs use both approaches to obtain comprehensive PK characterization.

